Solid emulsion definition chemistry9/18/2023  
Surfactants are usually organic compounds that are akin to amphiphilic, which means that this molecule, being as double-agent, each contains a hydrophilic "water-seeking" group (the head), and a hydrophobic "water-avoiding" group (the tail). When the droplet is aprotic it is sometimes known as a reverse micelle. 
The compounds that coat a micelle are typically amphiphilic in nature, meaning that micelles may be stable either as droplets of aprotic solvents such as oil in water, or as protic solvents such as water in oil. This inhibits the oil droplets, the hydrophobic cores of micelles, from merging into fewer, larger droplets ("emulsion breaking") of the micelle. The polar "heads" of the surfactant molecules coating the micelle interact more strongly with water, so they form a hydrophilic outer layer that forms a barrier between micelles. It is also recognized that in a stabilized emulsion done by a surfactant, the surfactant monomers surround the droplets like a thin film and prevents them from the mechanism of coalescence.Schematic diagram of a micelle – the lipophilic tails of the surfactant ions remain inside the oil because they interact more strongly with oil than with water. Based on the revised articles, it was found that emulsions stability is related to so many aspects such as phase ratios, surfactant concentrations, agitation, temperature, and other existing compounds in the system. This evaluation focused on the study of emulsion types, stability of the emulsion, and stability mechanisms. A vast number of products from food to cosmetics deal with emulsions such as mayonnaise, milk, butter, creams, shampoos, and so many other daily products. ConclusionĮmulsions are considered very essential in everyday life. Viscosity modification – Certain emulgents such as acacia, tragacanth, carboxymethylcellulose, polyethylene glycol, etc raise the viscosity of the medium, which helps to develop and maintain the suspension of globules of the dispersed phase. This repulsive force produces them to remain suspended in the dispersion medium. Repulsion theory – The theory states that the emulsifying agent makes a film over one of the phases that result in the formation of globules that repel each other. Surface tension theory – According to the theory of surface tension, emulsification takes place through the reduction of the interfacial tension that exists in the middle of the two phases. Several various chemical and physical processes and mechanisms can be contained in the procedure of emulsification. This is also known as the Tyndall effect. If the emulsion is dilute, then higher-frequency and the low-wavelength type of light will be scattered in more fractions, and this type of emulsion will be converted to a blue color.Emulsions occur in white color when the light is dispersed in equal proportions.Emulsions have a cloudy presence due to so many phase interfaces scattering light moving through the emulsions.a continuous and the dispersed with the boundary coming in between the phases, that is called “interface”. Emulsions contain both immiscible liquid phases i.e.Some of the most common examples of emulsifiers are egg yolk, mustard, sodium phosphates, DATEM, Mono and diglycerides, cellulose, soy lecithin, etc.Some commonly used emulsifiers for the Water in oil emulsions are the long-chain alcohols, heavy metal salts of fatty acids, etc.The generally used emulsifiers for Oil in water emulsions are proteins, gums, soaps, etc.On the other hand, if the emulsifier is more soluble in oil, then oil develops into the dispersion medium and water turns into the dispersed phase.If the emulsifier is more soluble in water then the water turn into the dispersion medium and oil is converted into the dispersed phase and hence we get oil in the water emulsion.It depends mainly on which amidst water and oil can solvate the emulsifier to a larger extent. It’s not just the percentage of water and oil that determines whether it is oil-in-water or a water-in-oil emulsion.Also, it can be cationic, anionic, or even non-polar. Emulsifiers build a layer between the dispersed phase and the dispersion medium, thereby preventing the dispersed phase particles to come into the league to build larger particles and separate. 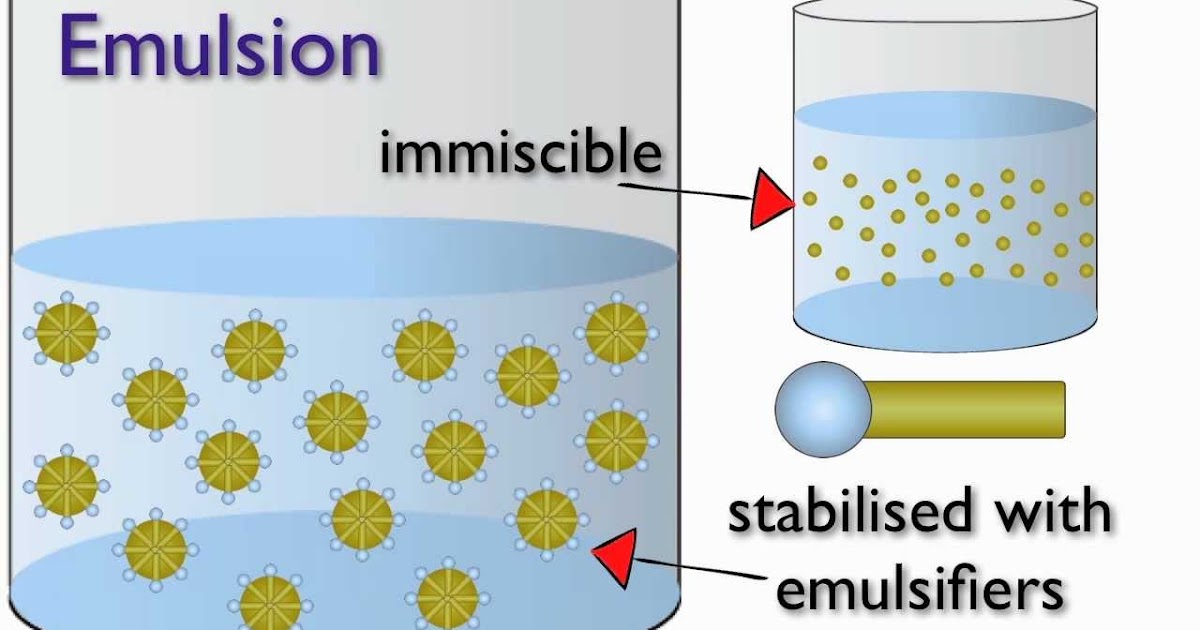
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
